Species of Thailand
Rock pigeon
Columba livia
Johann Friedrich Gmelin, 1789
In Thai: นกพิราบป่า
The rock dove, rock pigeon, or common pigeon ( also ; Columba livia) is a member of the bird family Columbidae (doves and pigeons). In common usage, this bird is often simply referred to as the "pigeon".
The domestic pigeon (Columba livia domestica, which includes about 1, 000 different breeds) descended from this species. Escaped domestic pigeons have increased the populations of feral pigeons around the world.
Wild rock doves are pale grey with two black bars on each wing, whereas domestic and feral pigeons vary in colour and pattern. Few differences are seen between males and females. The species is generally monogamous, with two squabs (young) per brood. Both parents care for the young for a time.
Habitats include various open and semi-open environments. Cliffs and rock ledges are used for roosting and breeding in the wild. Originally found wild in Europe, North Africa, and western Asia, pigeons have become established in cities around the world. The species is abundant, with an estimated population of 17 to 28 million feral and wild birds in Europe alone and up to 120 million worldwide.
Taxonomy and systematics
The rock dove was first described by German naturalist Johann Gmelin in 1789. The genus name Columba is the Latin word meaning "pigeon, dove", whose older etymology comes from the Ancient Greek κόλυμβος (kolumbos), "a diver", from κολυμβάω (kolumbao), "dive, plunge headlong, swim". Aristophanes (Birds, 304) and others use the word κολυμβίς (kolumbis), "diver", for the name of the bird, because of its swimming motion in the air. The specific epithet is derived from the Latin livor, "bluish". Its closest relative in the genus Columba is the hill pigeon, followed by the other rock pigeons: the snow, speckled, and white-collared pigeons.
The official common name is rock dove, as given by the International Ornithological Congress. In common usage, this bird is still often simply referred to as the "pigeon". Pigeon chicks are called squabs.
The rock dove was central to Charles Darwins discovery of evolution, and featured in four of his works from 1859 to 1872. Darwin posited, that despite wide ranging morphological differences, the many hundreds of breeds of domestic pigeons could all be traced back to the wild rock dove; in essence human selection of pigeon breeds was analogous to natural selection.
Subspecies
Twelve subspecies are recognised by Gibbs (2000); some of these may be derived from feral stock.
- The European rock dove (C. l. livia) – The nominate subspecies; it occurs in western and southern Europe, northern Africa, and Asia to western Kazakhstan, the northern Caucasus, Georgia, Cyprus, Turkey, Iran, and Iraq.
- The Cape Verde rock dove (C. l. atlantis) (Bannerman, 1931) – Found in Madeira, the Azores and Cape Verde, it is a very variable population with chequered upperparts obscuring the black wingbars, and is almost certainly derived from feral pigeons.
- The Canary Islands rock dove (C. l. canariensis) (Bannerman, 1914) – Found in the Canary Islands, it is smaller and is usually darker than the nominate subspecies.
- The Senegal rock dove (C. l. gymnocyclus) (Gray, 1856) – Found from Senegal and Guinea to Ghana, Benin and Nigeria, it is smaller and very much darker than the nominate. It is almost blackish on the head, rump, and underparts with a white back and the iridescence of the nape extending onto the head.
- The Saharan rock dove (C. l. targia) (Geyr von Schweppenburg, 1916) – It breeds in the mountains of the Sahara east to Sudan. It is slightly smaller than the nominate form, with similar plumage, but the back is concolorous with the mantle instead of white.
- The oasis rock dove (C. l. dakhlae) (Richard Meinertzhagen, 1928) – Found only in two oases in central Egypt, it is smaller and much paler than the nominate subspecies.
- The Egyptian rock dove (C. l. schimperi) (Bonaparte, 1854) – Found in the Nile Delta south to northern Sudan, it closely resembles C. l. targia, but has a distinctly paler mantle.
- The Arabian rock dove (C. l. palaestinae) (Zedlitz, 1912) – Ranging from Syria to Sinai and Arabia, it is slightly larger than C. l. schimperi and has darker plumage.
- The Iranian rock dove (C. l. gaddi) (Zarodney & Looudoni, 1906) – Breeds from Azerbaijan and Iran east to Uzbekistan and is larger and paler than C. l. palaestinae, with which it intergrades in the west. It also intergrades with the next subspecies to the east.
- Hume's rock dove (C. l. neglecta) (Hume, 1873) – Found in the mountains of eastern Central Asia, it is similar to the nominate subspecies in size, but is darker with a stronger and more extensive iridescent sheen on the neck. It intergrades with the next race in the south.
- The Indian rock dove (C. l. intermedia) (Strickland, 1844) – Occurs in Sri Lanka and in India south of the Himalayan range of C. l. neglecta. It is similar to that subspecies, but darker with a less contrasting back.
- The Mongolian rock dove (C. l. nigricans) (Buturlin, 1908) – Found in Mongolia and northern China, it is variable and probably derived from feral pigeons.
Description
The adult of the nominate subspecies of the rock dove is 29 to 37 cm long with a 62 to 72 cm wingspan. Weight for wild or feral rock doves ranges from 238 - 380 g abbr = on, though overfed domestic and semidomestic individuals can exceed normal weights. It has a dark bluish-grey head, neck, and chest with glossy yellowish, greenish, and reddish-purple iridescence along its neck and wing feathers. The iris is orange, red, or golden with a paler inner ring, and the bare skin round the eye is bluish-grey. The bill is grey-black with a conspicuous off-white cere, and the feet are purplish-red. Among standard measurements, the wing chord is typically around 22.3 cm, the tail is 9.5 to 11 cm, the bill is around 1.8 cm, and the tarsus is 2.6 to 3.5 cm.
The adult female is almost identical in outward appearance to the male, but the iridescence on her neck is less intense and more restricted to the rear and sides, whereas that on the breast is often very obscure.
The white lower back of the pure rock dove is its best identification characteristic; the two black bars on its pale grey wings are also distinctive. The tail has a black band on the end, and the outer web of the tail feathers are margined with white. It is strong and quick on the wing, dashing out from sea caves, flying low over the water, its lighter grey rump showing well from above.
Young birds show little lustre and are duller. Eye colour of the pigeon is generally orange, but a few pigeons may have white-grey eyes. The eyelids are orange and encapsulated in a grey-white eye ring. The feet are red to pink.
When circling overhead, the white underwing of the bird becomes conspicuous. In its flight, behaviour, and voice, which is more of a dovecot coo than the phrase of the wood pigeon, it is a typical pigeon. Although it is a relatively strong flier, it also glides frequently, holding its wings in a very pronounced V shape as it does. As prey birds, they must keep their vigilance, and when disturbed a pigeon within a flock will take off with a noisy clapping sound that cues for other pigeons to take to flight. The noise of the take-off increases the faster a pigeon beats its wings, thus advertising the magnitude of a perceived threat to its flockmates.
Pigeons feed on the ground in flocks or individually. Pigeons are naturally granivorous, eating seeds that fit down their gullet. They may sometimes consume small invertebrates such as worms or insect larvae as a protein supplement. As they do not possess an enlarged cecum as in European wood pigeons, they cannot digest adult plant tissue; the various seeds they eat containing the appropriate nutrients they require. While most birds take small sips and tilt their heads backwards when drinking, pigeons are able to dip their bills into the water and drink continuously, without having to tilt their heads back. In cities they typically resort to scavenging human garbage, as unprocessed grain may be impossible to find. Pigeon groups typically consist of producers, which locate and obtain food, and scroungers, which feed on food obtained by the producers. Generally, groups of pigeons contain a greater proportion of scroungers than producers.
Pigeons, especially homing or carrier breeds, are well known for their ability to find their way home from long distances. Despite these demonstrated abilities, wild rock doves are sedentary and rarely leave their local areas.
A rock pigeon's lifespan ranges from 3–5 years in the wild to 15 years in captivity, though longer-lived specimens have been reported. The main causes of mortality in the wild are predators and persecution by humans. The species was first introduced to North America in 1606 at Port Royal, Nova Scotia.
Distribution and habitat
The rock dove has a restricted natural resident range in western and southern Europe, North Africa, and extending into South Asia. It is often found in pairs in the breeding season, but is usually gregarious. The species (including ferals) has a large range, with an estimated global extent of occurrence of 10000000 km2. It has a large global population, including an estimated 17 to 28 million individuals in Europe. Fossil evidence suggests the rock dove originated in southern Asia, and skeletal remains, unearthed in Israel, confirm its existence there for at least three-hundred thousand years. However, this species has such a long history with humans that it is impossible to identify the species' original range exactly.
Pigeons in the wild reside in rock formations, settling in crevices to nest. They nest communally, often forming large colonies of many hundreds of individuals. Feral pigeons are usually unable to find these accommodations, so they must nest on building ledges, walls or statues. They may damage these structures via their feces; starving birds can only excrete urates, which over time corrodes masonry and metal. In contrast, a well-fed bird passes mostly solid feces, containing only small amounts of uric acid.
Breeding
The rock dove breeds at any time of the year, but peak times are spring and summer. Nesting sites are along coastal cliff faces, as well as the artificial cliff faces created by apartment buildings with accessible ledges or roof spaces.
The nest is a flimsy platform of straw and sticks, laid on a ledge, under cover, often on the window ledges of buildings. Two white eggs are laid; incubation, shared by both parents, lasts 17 to 19 days. The newly hatched squab (nestling) has pale yellow down and a flesh-coloured bill with a dark band. For the first few days, the baby squabs are tended and fed (through regurgitation) exclusively on "crop milk" (also called "pigeon milk" or "pigeon's milk"). The pigeon milk is produced in the crops of both parents in all species of pigeons and doves. The fledging period is about 30 days.
Feeding
Rock doves are omnivorous, but prefer plant matter: chiefly fruits and grains.
Preening
Pigeons primarily use powder down feathers for preening, which gives a soft and silky feel to their plumage. They have no preen gland or at times have very rudimentary preen glands, so oil is not used for preening. Rather, powder down feathers are spread across the body. These have a tendency to disintegrate, and the powder, akin to talcum powder, helps maintain the plumage. Some varieties of domestic pigeons have modified feathers called "fat quills". These feathers contain yellow, oil-like fat that derives from the same cells as powder down. This is used while preening and helps reduce bacterial degradation of feathers by feather bacilli.
Predators
With only their flying abilities protecting them from predation, rock pigeons are a favourite almost around the world for a wide range of raptors. In fact, with feral pigeons existing in almost every city in the world, they may form the majority of prey for several raptor species that live in urban areas. Peregrine falcons and Eurasian sparrowhawks are natural predators of pigeons that are quite adept at catching and feeding upon this species. Up to 80% of the diet of peregrine falcons in several cities that have breeding falcons is composed of feral pigeons. Some common predators of feral pigeons in North America are opossums, raccoons, red-tailed hawks, great horned owls, eastern screech owls, and accipiters. The birds that prey on pigeons in North America can range in size from American kestrels to golden eagles and can even include gulls, crows, and ravens. On the ground, the adults, their young, and their eggs are at risk from feral and domestic cats. Doves and pigeons are considered to be game birds, as many species have been hunted and used for food in many of the countries in which they are native.
The body feathers have dense, fluffy bases and are loosely attached to the skin, hence they drop out easily. When a predator catches it, large numbers of feathers come out in the attacker's mouth, and the pigeon may use this temporary distraction to make an escape. It also tends to drop the tail feathers when preyed upon or under traumatic conditions, probably as a distraction mechanism.
Parasites
| Tinaminyssus melloi, a nasal mite. | Pigeon louse fly (Pseudolynchia canariensis), a blood-sucking ectoparasite. |
Pigeons may harbour a diverse parasite fauna.
They often host the intestinal helminths Capillaria columbae and Ascaridia columbae. Their ectoparasites include the ischnoceran lice Columbicola columbae, Campanulotes bidentatus compar, the amblyceran lice Bonomiella columbae, Hohorstiella lata, Colpocephalum turbinatum, the mites Tinaminyssus melloi, Dermanyssus gallinae, Dermoglyphus columbae, Falculifer rostratus, and Diplaegidia columbae. The hippoboscid fly Pseudolynchia canariensis is a typical blood-sucking ectoparasite of pigeons, found only in tropical and subtropical regions.
Domestication
Rock doves have been domesticated for several thousand years, giving rise to the domestic pigeon (Columba livia domestica). They may have been domesticated as many as 5, 000 years ago. Numerous breeds of fancy pigeons of all sizes, colours, and types have been bred. Domesticated pigeons are used as homing pigeons as well as food and pets. They were in the past also used as carrier pigeons, and so-called war pigeons have played significant roles during wartime, including delivering urgent medicines, with many pigeons having received bravery awards and medals for their services in saving hundreds of human lives, including, notably, the British pigeon Cher Ami, which received the Croix de Guerre for actions during World War I, and the Irish Paddy and the American G.I. Joe, which both received the Dickin Medal, amongst 32 pigeons to receive this award, for their actions during World War II, more than any other species of animal
Feral pigeon
Many domestic birds have got lost, escaped or been released over the years, and have given rise to feral pigeons. These show a variety of plumages, although many have the blue-barred pattern as does the pure rock dove. Feral pigeons are found in cities and towns all over the world. The scarcity of the pure wild species is partly due to interbreeding with feral birds.
Human health
Contact with pigeon droppings poses a minor risk of contracting histoplasmosis, cryptococcosis, and psittacosis, and long-term exposure to both droppings and feathers can induce an allergy known as bird fancier's lung.
Pigeons are not a major concern in the spread of West Nile virus; though they can contract it, they do not appear to be able to transmit it. Pigeons are, however, at potential risk for carrying and spreading avian influenza. One study has shown that adult pigeons are not clinically susceptible to the most dangerous strain of avian influenza, H5N1, and that they did not transmit the virus to chickens. Other studies have presented evidence of clinical signs and neurological lesions resulting from infection, but found that the pigeons did not transmit the disease to chickens reared in direct contact with them. Pigeons were found to be "resistant or minimally susceptible" to other strains of avian influenza, such as the H7N7.
This article uses material from Wikipedia released under the Creative Commons Attribution-Share-Alike Licence 3.0. Eventual photos shown in this page may or may not be from Wikipedia, please see the license details for photos in photo by-lines.
Category / Seasonal Status
Wiki listed status (concerning Thai population): Due to hybridisation with feral pigeons, few if any pure-bred birds remain in Thailand
BCST Category: Introduced, with feral breeding stock apparently self-supporting
BCST Seasonal status: Resident or presumed resident
Scientific classification
- Kingdom
- Animalia
- Phylum
- Chordata
- Class
- Aves
- Order
- Columbiformes
- Family
- Columbidae
- Genus
- Columba
- Species
- Columba livia
Common names
- English:
- Common pigeon
- Rock dove
- Rock doveI
- French: Pigeon biset
- Thai: นกพิราบป่า
Conservation status

Least Concern (IUCN3.1)
Photos
Please help us review the bird photos if wrong ones are used. We can be reached via our contact us page.
Range Map
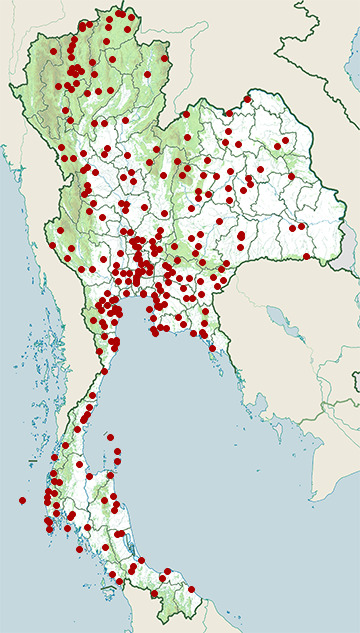
- Amphawa District, Samut Songkhram
- Ao Manao-Khao Tanyong National Park
- Ao Phang-Nga National Park
- Aranyaprathet District, Sa Kaeo
- Ban Bueng District, Chonburi
- Ban Chang District, Rayong
- Ban Laem District, Phetchaburi
- Ban Lat District, Phetchaburi
- Ban Phai District, Khon Kaen
- Ban Pho District, Chachoengsao
- Ban Phraek District, Phra Nakhon Si Ayutthaya
- Ban Sang District, Prachinburi
- Bang Ban District, Phra Nakhon Si Ayutthaya
- Bang Kruai District, Nonthaburi
- Bang Lamung District, Chonburi
- Bang Len District, Nakhon Pathom
- Bang Pa In District, Phra Nakhon Si Ayutthaya
- Bang Pahan District, Phra Nakhon Si Ayutthaya
- Bang Pakong District, Chachoengsao
- Bang Phra Non-Hunting Area
- Bang Pu Recreation Centre
- Bang Saphan Noi District, Prachuap Khiri Khan
- Bangkok Province
- Borabue District, Maha Sarakham
- Bueng Boraped Non-Hunting Area
- Chaiya District, Surat Thani
- Chaiyo District, Ang Thong
- Chaloem Phra Kiat District, Saraburi
- Chaloem Phrakiat Thai Prachan National Park
- Chatturat District, Chaiyaphum
- Chiang Dao District, Chiang Mai
- Chiang Dao Wildlife Sanctuary
- Chiang Khan District, Loei
- Chiang Khong District, Chiang Rai
- Chiang Saen District, Chiang Rai
- Doi Inthanon National Park
- Doi Lo District, Chiang Mai
- Doi Pha Hom Pok National Park
- Doi Saket District, Chiang Mai
- Doi Suthep - Pui National Park
- Doi Tao District, Chiang Mai
- Fang District, Chiang Mai
- Hang Chat District, Lampang
- Hat Wanakon National Park
- Hat Yai District, Songkhla
- Hua Hin District, Prachuap Khiri Khan
- Huai Chorakhe Mak Reservoir Non-Hunting Area
- Huai Kha Khaeng Wildlife Sanctuary
- Huai Krachao District, Kanchanaburi
- Huai Talat Reservoir Non-Hunting Area
- In Buri District, Sing Buri
- Kabin Buri District, Prachinburi
- Kaeng Khoi District, Saraburi
- Kaeng Krachan District, Phetchaburi
- Kaeng Krachan National Park
- Kamphaeng Saen District, Nakhon Pathom
- Kanthararom District, Sisaket
- Kantharawichai District, Maha Sarakham
- Kapong District, Phang Nga
- Khanom District, Nakhon Si Thammarat
- Khao Ang Rue Nai Wildlife Sanctuary
- Khao Dinsor (Chumphon Raptor Center)
- Khao Khiao - Khao Chomphu Wildlife Sanctuary
- Khao Khitchakut National Park
- Khao Laem National Park
- Khao Lak - Lam Ru National Park
- Khao Luang National Park
- Khao Nang Phanthurat Forest Park
- Khao Phanom Bencha National Park
- Khao Phra - Bang Khram Wildlife Sanctuary
- Khao Phra Wihan National Park
- Khao Sam Roi Yot National Park
- Khao Sanam Prieng Wildlife Sanctuary
- Khao Soi Dao Wildlife Sanctuary
- Khao Sok National Park
- Khao Yai National Park
- Khao Yoi District, Phetchaburi
- Khlong Hoi Khong District, Songkhla
- Khlong Lan National Park
- Khlong Luang District, Pathum Thani
- Khlong Wang Chao National Park
- Khok Pho District, Pattani
- Khok Sung District, Sa Kaeo
- Khon San District, Chaiyaphum
- Khuan Khanun District, Phatthalung
- Khun Chae National Park
- Khun Nan National Park
- Khun Phawo National Park
- Khun Tan District, Chiang Rai
- Khung Kraben Non-Hunting Area
- Khura Buri District, Phang Nga
- Klaeng District, Rayong
- Ko Lanta National Park
- Ko Samui District, Surat Thani
- Ko Sichang District, Chonburi
- Ko Tao
- Kui Buri National Park
- Kumphawapi District, Udon Thani
- Laem Ngop District, Trat
- Laem Pak Bia
- Laem Son National Park
- Mae Ai District, Chiang Mai
- Mae Chan District, Chiang Rai
- Mae Mo District, Lampang
- Mae Ping National Park
- Mae Poen District, Nakhon Sawan
- Mae Rim District, Chiang Mai
- Mae Sot District, Tak
- Mae Taeng District, Chiang Mai
- Mae Wong National Park
- Mueang Buriram District, Buriram
- Mueang Chachoengsao District, Chachoengsao
- Mueang Chaiyaphum District, Chaiyaphum
- Mueang Chanthaburi District, Chanthaburi
- Mueang Chiang Mai District, Chiang Mai
- Mueang Chiang Rai District, Chiang Rai
- Mueang Chonburi District, Chonburi
- Mueang Chumphon District, Chumphon
- Mueang Kalasin District, Kalasin
- Mueang Kamphaeng Phet District, Kamphaeng Phet
- Mueang Kanchanaburi District, Kanchanaburi
- Mueang Khon Kaen District, Khon Kaen
- Mueang Krabi District, Krabi
- Mueang Lampang District, Lampang
- Mueang Lamphun District, Lamphun
- Mueang Lopburi District, Lopburi
- Mueang Maha Sarakham District, Maha Sarakham
- Mueang Nakhon Nayok District, Nakhon Nayok
- Mueang Nakhon Pathom District, Nakhon Pathom
- Mueang Nakhon Ratchasima District, Nakhon Ratchasima
- Mueang Nakhon Sawan District, Nakhon Sawan
- Mueang Nakhon Si Thammarat District, Nakhon Si Thammarat
- Mueang Nan District, Nan
- Mueang Nong Khai District, Nong Khai
- Mueang Nonthaburi District, Nonthaburi
- Mueang Pan District, Lampang
- Mueang Pathum Thani District, Pathum Thani
- Mueang Pattani District, Pattani
- Mueang Phang Nga District, Phang Nga
- Mueang Phayao District, Phayao
- Mueang Phetchabun District, Phetchabun
- Mueang Phetchaburi District, Phetchaburi
- Mueang Phichit District, Phichit
- Mueang Phitsanulok District, Phitsanulok
- Mueang Phuket District, Phuket
- Mueang Prachinburi District, Prachinburi
- Mueang Ratchaburi District, Ratchaburi
- Mueang Rayong District, Rayong
- Mueang Sa Kaeo District, Sa Kaeo
- Mueang Samut Sakhon District, Samut Sakhon
- Mueang Samut Songkhram District, Samut Songkhram
- Mueang Saraburi District, Saraburi
- Mueang Satun District, Satun
- Mueang Sisaket District, Sisaket
- Mueang Songkhla District, Songkhla
- Mueang Sukhothai District, Sukhothai
- Mueang Suphanburi District, Suphan Buri
- Mueang Surat Thani District, Surat Thani
- Mueang Tak District, Tak
- Mueang Udon Thani District, Udon Thani
- Mueang Uttaradit District, Uttaradit
- Nam Nao National Park
- Nam Phong National Park
- Namtok Phlio National Park
- Ngao Waterfall National Park
- Non Din Daeng District, Buriram
- Nong Bong Khai Non-Hunting Area
- Nong Han Lake
- Nong Song Hong District, Khon Kaen
- Nong Suea District, Pathum Thani
- Nong Waeng Non-Hunting Area
- Nong Ya Plong District, Phetchaburi
- Nong Yai Area Development Project Under Royal Init
- Pa Sak Chonlasit Dam Non-Hunting Area
- Pa Sang District, Lamphun
- Pai District, Mae Hong Son
- Pak Chong District, Nakhon Ratchasima
- Pak Kret District, Nonthaburi
- Pak Phli District, Nakhon Nayok
- Pak Thale
- Pak Tho District, Ratchaburi
- Pak Thong Chai District, Nakhon Ratchasima
- Pang Sila Thong District, Kamphaeng Phet
- Pathio District, Chumphon
- Pha Daeng National Park
- Pha Nam Yoi Forest Park
- Phaisali District, Nakhon Sawan
- Phan District, Chiang Rai
- Phanat Nikhom District, Chonburi
- Phatthana Nikhom District, Lopburi
- Phayuha Khiri District, Nakhon Sawan
- Phi Phi Islands
- Phimai District, Nakhon Ratchasima
- Pho Prathap Chang District, Phichit
- Phra Nakhon Si Ayutthaya District, Phra Nakhon Si Ayutthaya
- Phra Phrom District, Nakhon Si Thammarat
- Phu Foi Lom National Park
- Phu Hin Rong Kla National Park
- Phu Khiao District, Chaiyaphum
- Phu Khiao Wildlife Sanctuary
- Phu Phan National Park
- Phu Wiang National Park
- Phutthamonthon District, Nakhon Pathom
- Pran Buri District, Prachuap Khiri Khan
- Pran Buri Forest Park
- Ramkhamhaeng National Park
- Ratchasan District, Chachoengsao
- Rattanawapi District, Nong Khai
- Sai Noi District, Nonthaburi
- Sai Yok District, Kanchanaburi
- Sai Yok National Park
- Sakaerat Environmental Research Station
- Samae San Island
- Samut Prakan Province
- San Kala Khiri National Park
- San Sai District, Chiang Mai
- Sanam Bin Reservoir Non-Hunting Area
- Sanam Chai Khet District, Chachoengsao
- Sankhaburi District, Chainat
- Sattahip District, Chonburi
- Sawi District, Chumphon
- Si Maha Phot District, Prachinburi
- Si Racha District, Chonburi
- Si Satchanalai District, Sukhothai
- Si Satchanalai National Park
- Si Thep District, Phetchabun
- Similan Islands
- Sirinat National Park
- Song Phi Nong District, Suphan Buri
- Sri Nakarin Dam National Park
- Sri Phang Nga National Park
- Sung Noen District, Nakhon Ratchasima
- Ta Phraya National Park
- Taksin Maharat National Park
- Takua Pa District, Phang Nga
- Taphan Hin District, Phichit
- Tat Ton National Park
- Tha Chang District, Sing Buri
- Tha Phae District, Satun
- Tha Sala District, Nakhon Si Thammarat
- Tha Takiap District, Chachoengsao
- Tha Wung District, Lopburi
- Tha Yang District, Phetchaburi
- Thai Mueang District, Phang Nga
- Thalang District, Phuket
- Thale Noi Non-Hunting Area
- Tham Pratun Non-Hunting Area
- Than Sadet - Koh Pha-Ngan National Park
- Thanyaburi District, Pathum Thani
- Thao Kosa Forest Park
- Thap Lan National Park
- Thong Pha Phum National Park
- Thung Salaeng Luang National Park
- Wang Chan District, Rayong
- Wang Nam Yen District, Sa Kaeo
- Wang Noi District, Phra Nakhon Si Ayutthaya
- Wang Saphung District, Loei
- Wat Phai Lom & Wat Ampu Wararam Non-Hunting Area
- Wat Tham Erawan Non-Hunting Area
- Watthana Nakhon District, Sa Kaeo
- Yan Ta Khao District, Trang



